By Mihaela Popoviciu, Lorena Paduraru, Galal Yahya , Kamel Metwally and Simona Cavalu
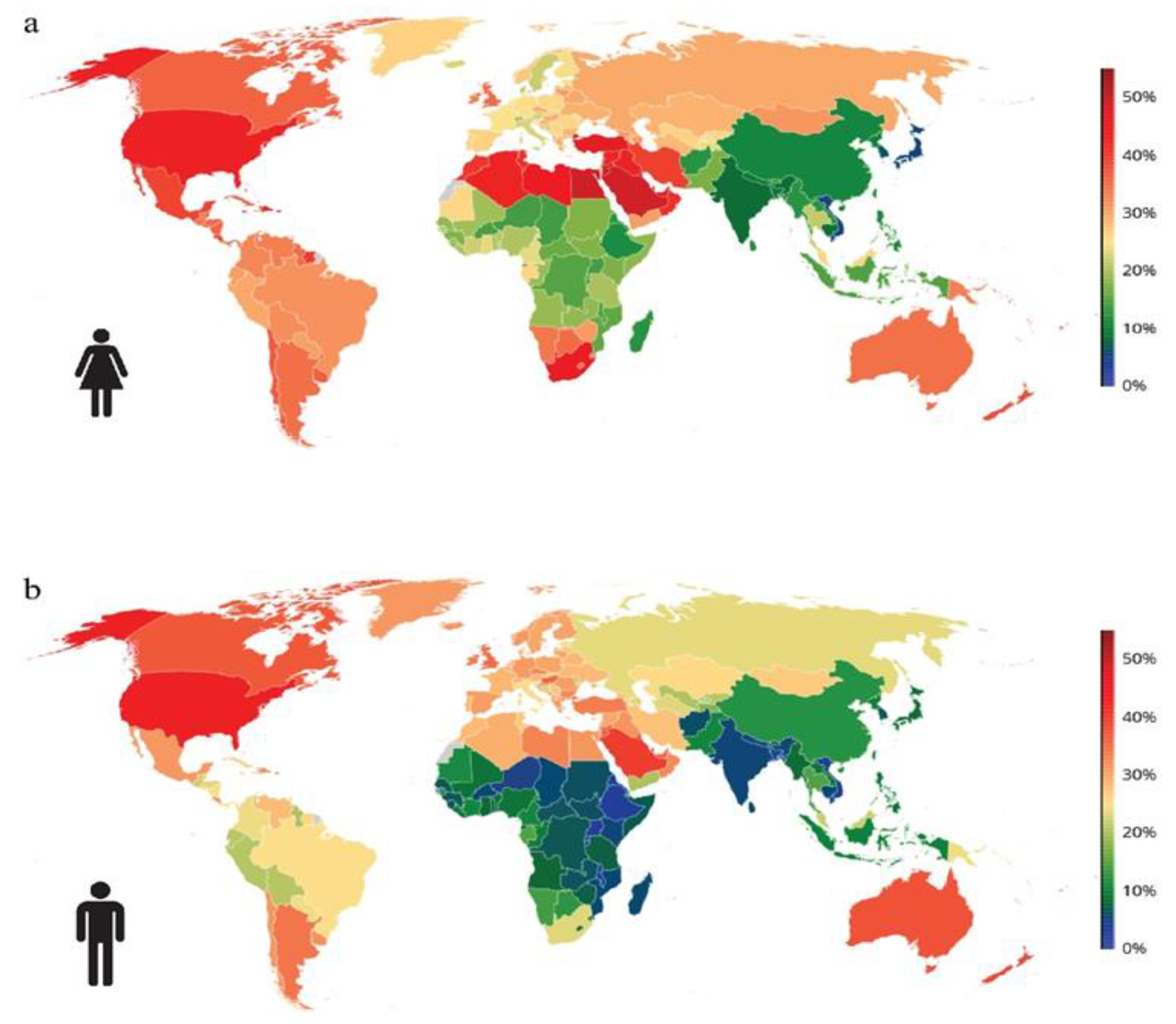
Obesity is a chronic disease with high prevalence and associated comorbidities, making it a growing global concern. These comorbidities include type 2 diabetes, hypertension, ventilatory dysfunction, arthrosis, venous and lymphatic circulation diseases, depression, and others, which have a negative impact on health and increase morbidity and mortality. GLP-1 agonists, used to treat type 2 diabetes, have been shown to be effective in promoting weight loss in preclinical and clinical studies. This review summarizes numerous studies conducted on the main drugs in the GLP-1 agonists class, outlining the maximum achievable weight loss. Our aim is to emphasize the active role and main outcomes of GLP-1 agonists in promoting weight loss, as well as in improving hyperglycemia, insulin sensitivity, blood pressure, cardio–metabolic, and renal protection. We highlight the pleiotropic effects of these medications, along with their indications, contraindications, and precautions for both diabetic and non-diabetic patients, based on long-term follow-up studies. Copyright Simona Cavalu et al.
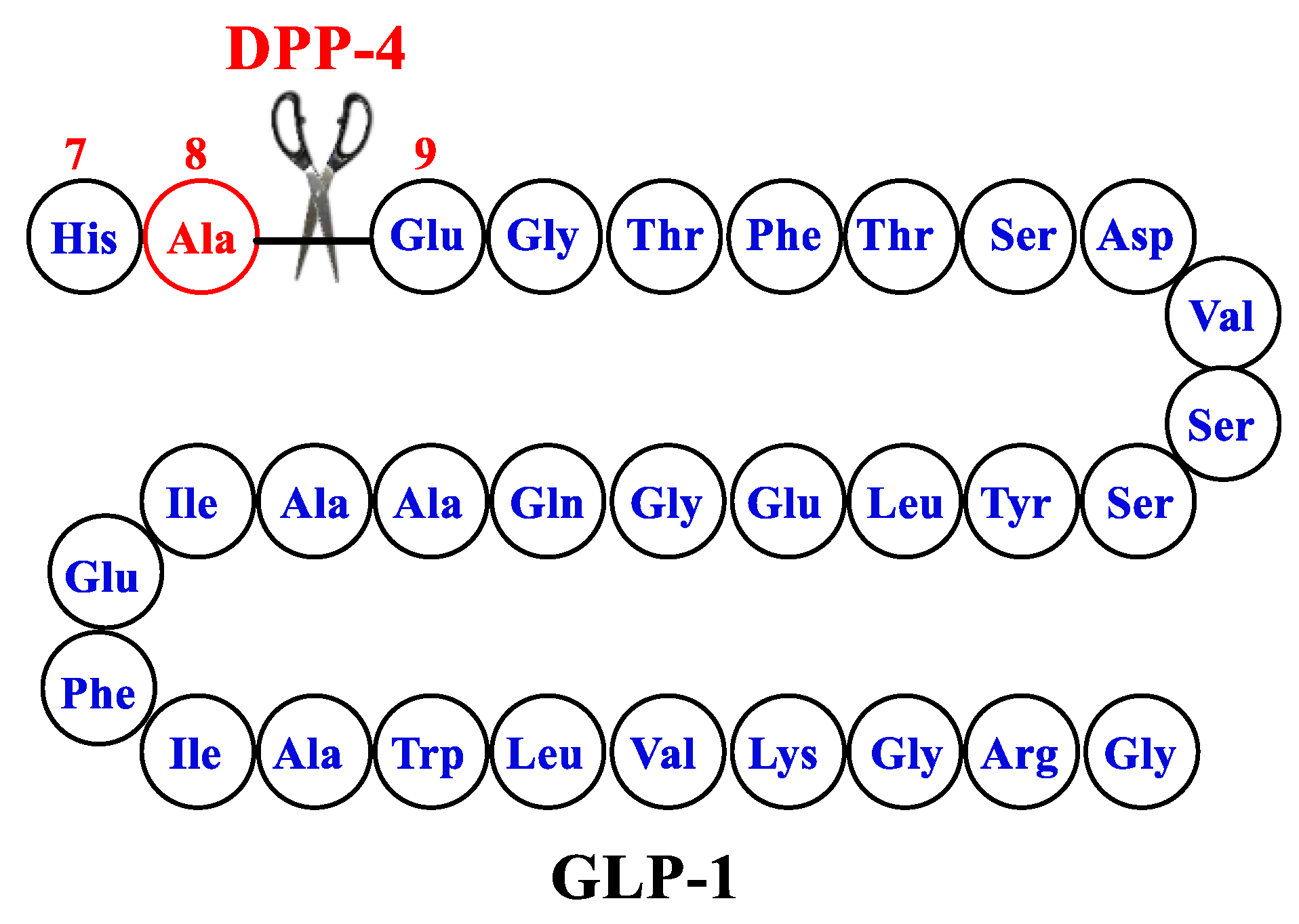
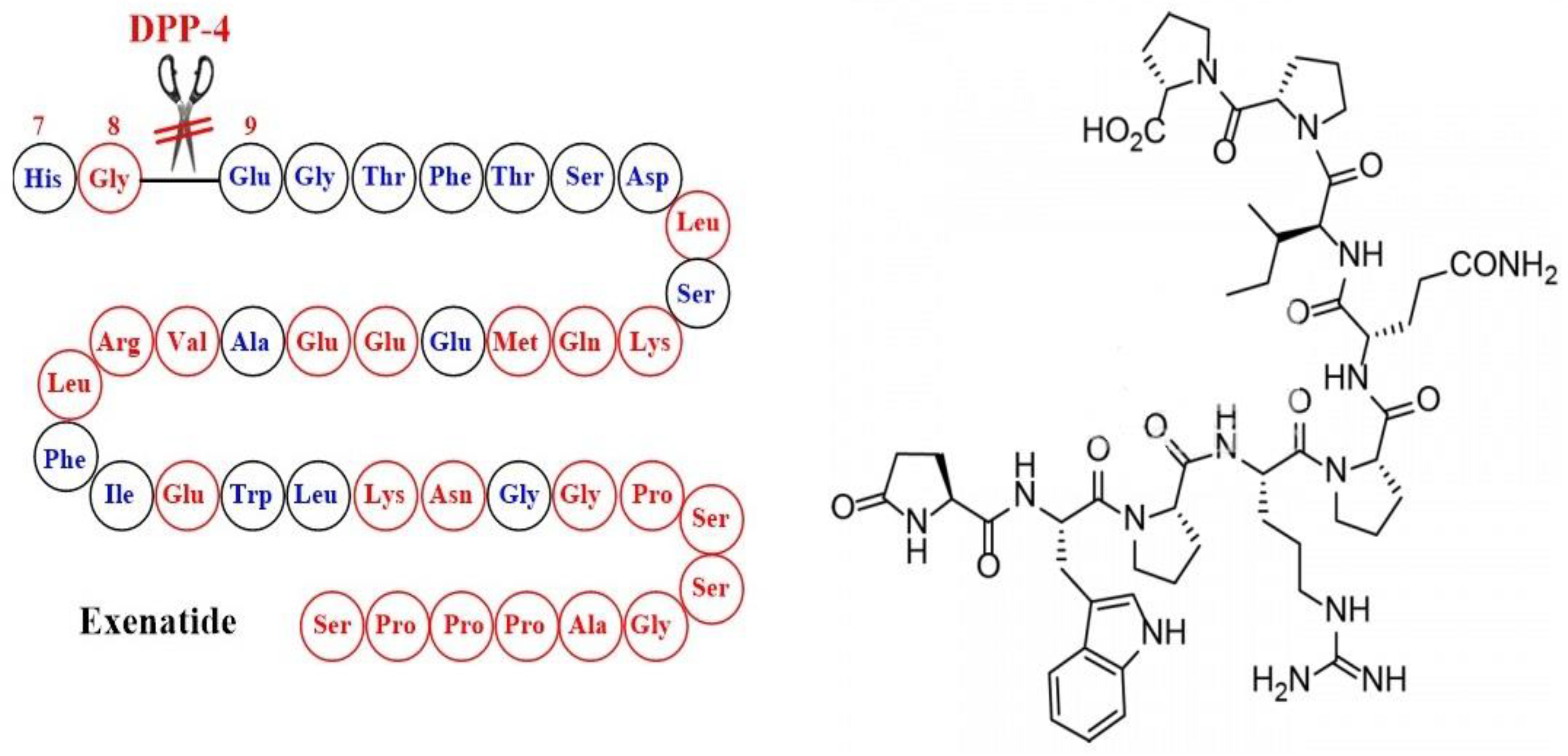
The Emergence of Glucagon-Like Peptide 1 Receptor Agonists and the First Results Obtained upon Administration of Exenatide. Copyright: Simona Cavalu etal.
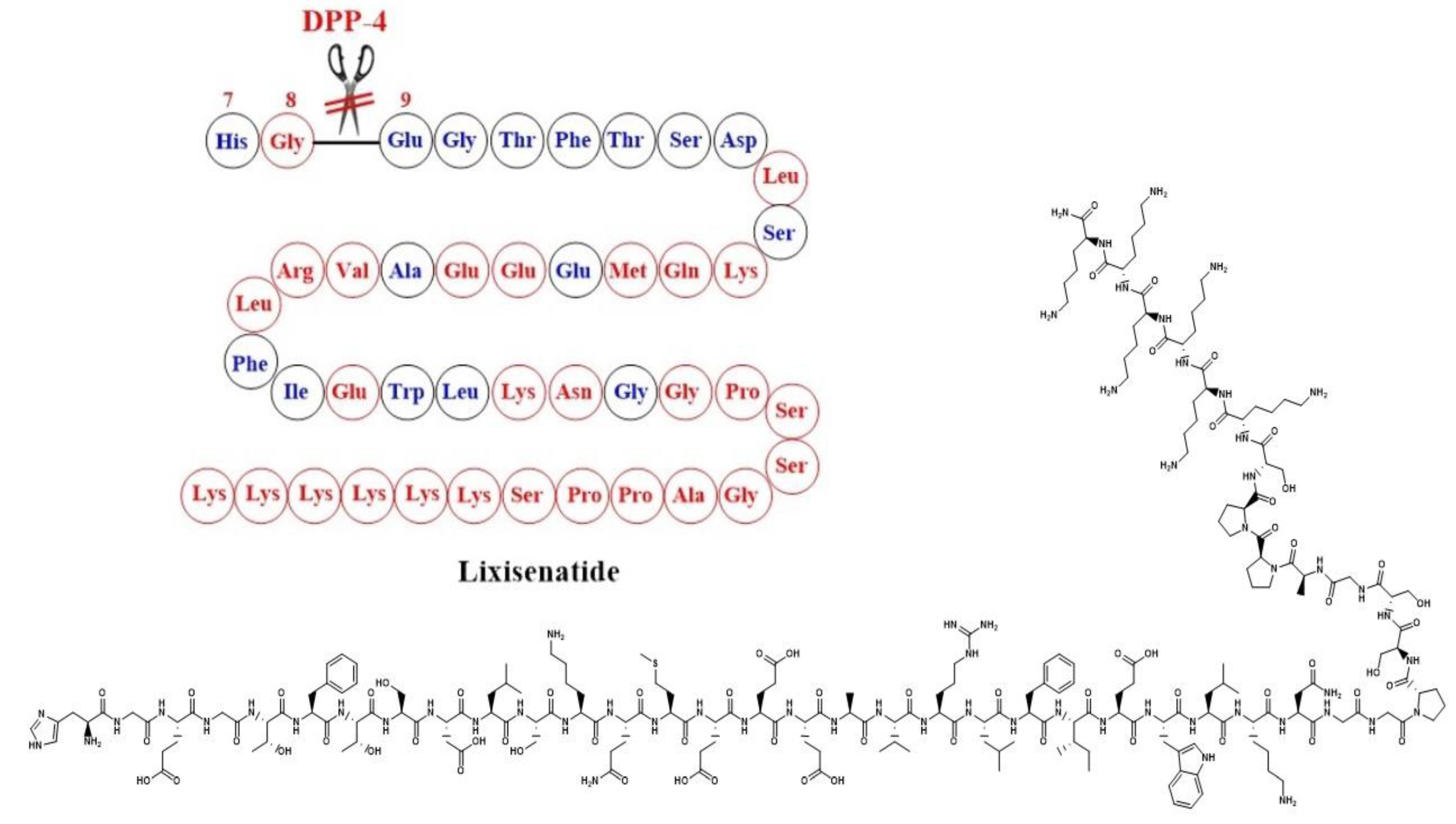
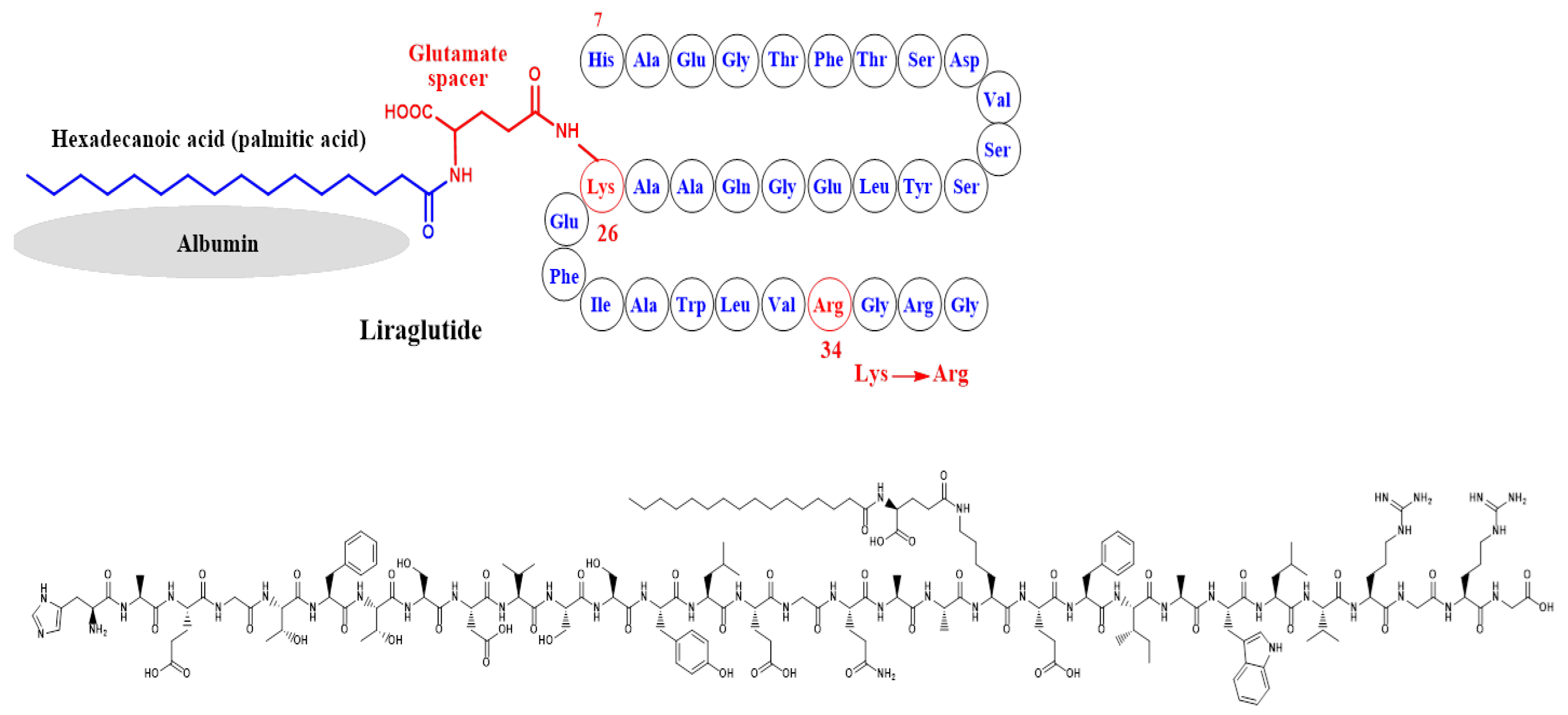
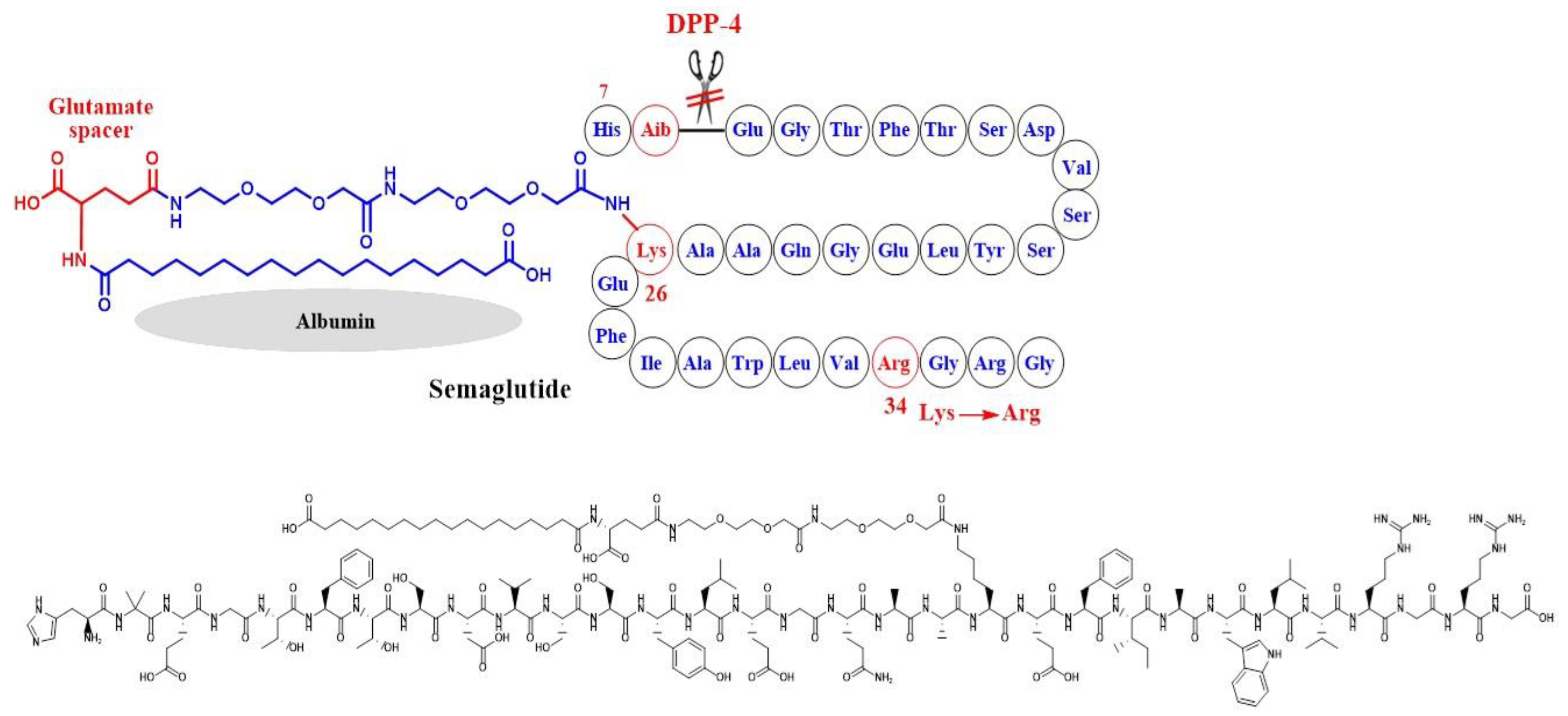
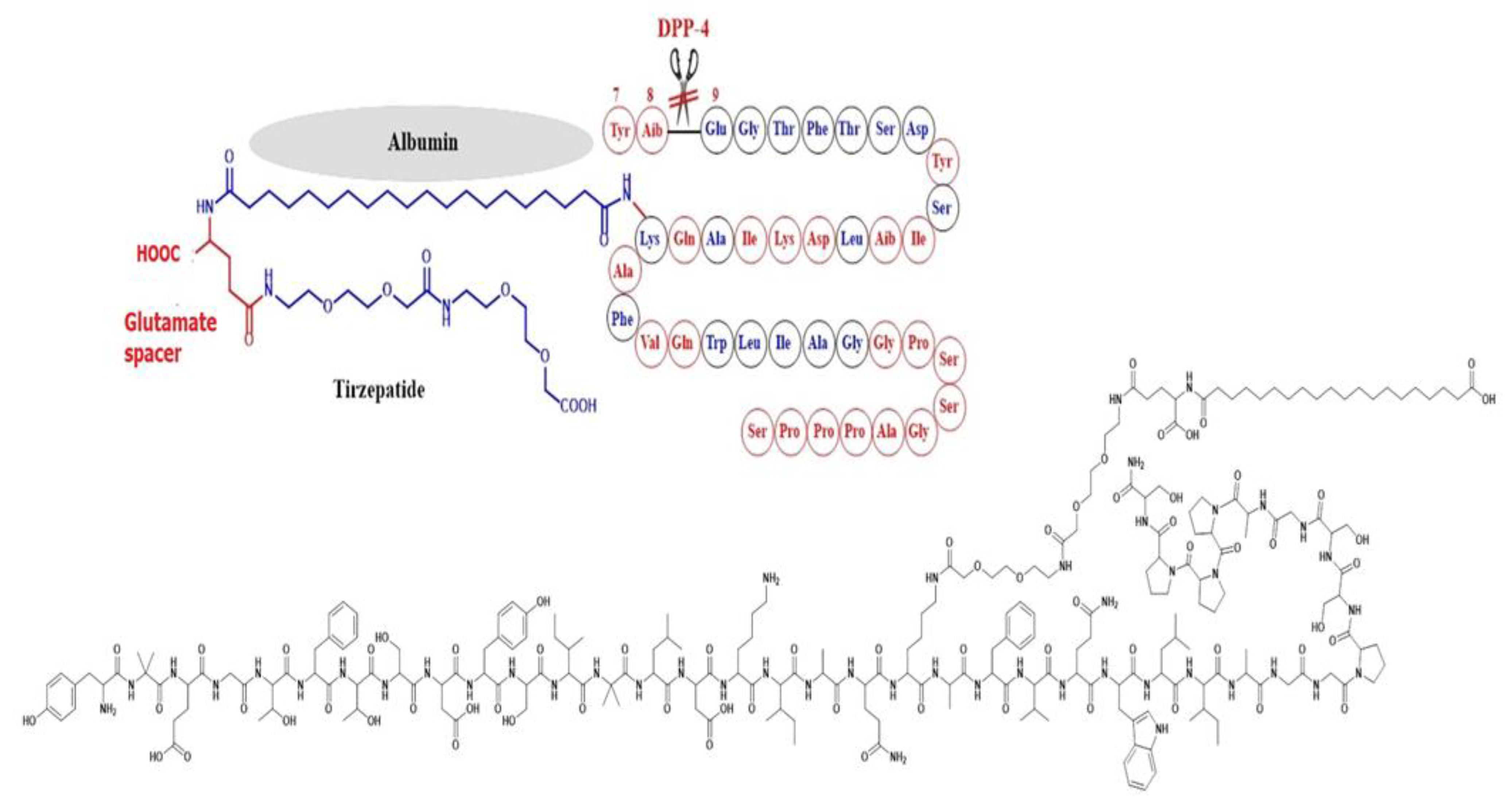
Simona Cavalu et al.
The combined central and peripheral actions of GLP-1 RA promote satiety, decrease hunger, and ultimately reduce food intake. While GLP-1 RA-induced deceleration of gastric emptying and occasional nausea may contribute to the weight-reducing effects, they appear to play a minor and temporary role. The inhibition of food intake by GLP-1/RA-mediated GLP-1 has been attributed to both direct central actions, with GLP-1 receptors present in brain regions involved in food intake and energy balance, and indirect pathways via vagal afferents originating in the gut and portal circulation. Copyright: Simona Cavalu et al.
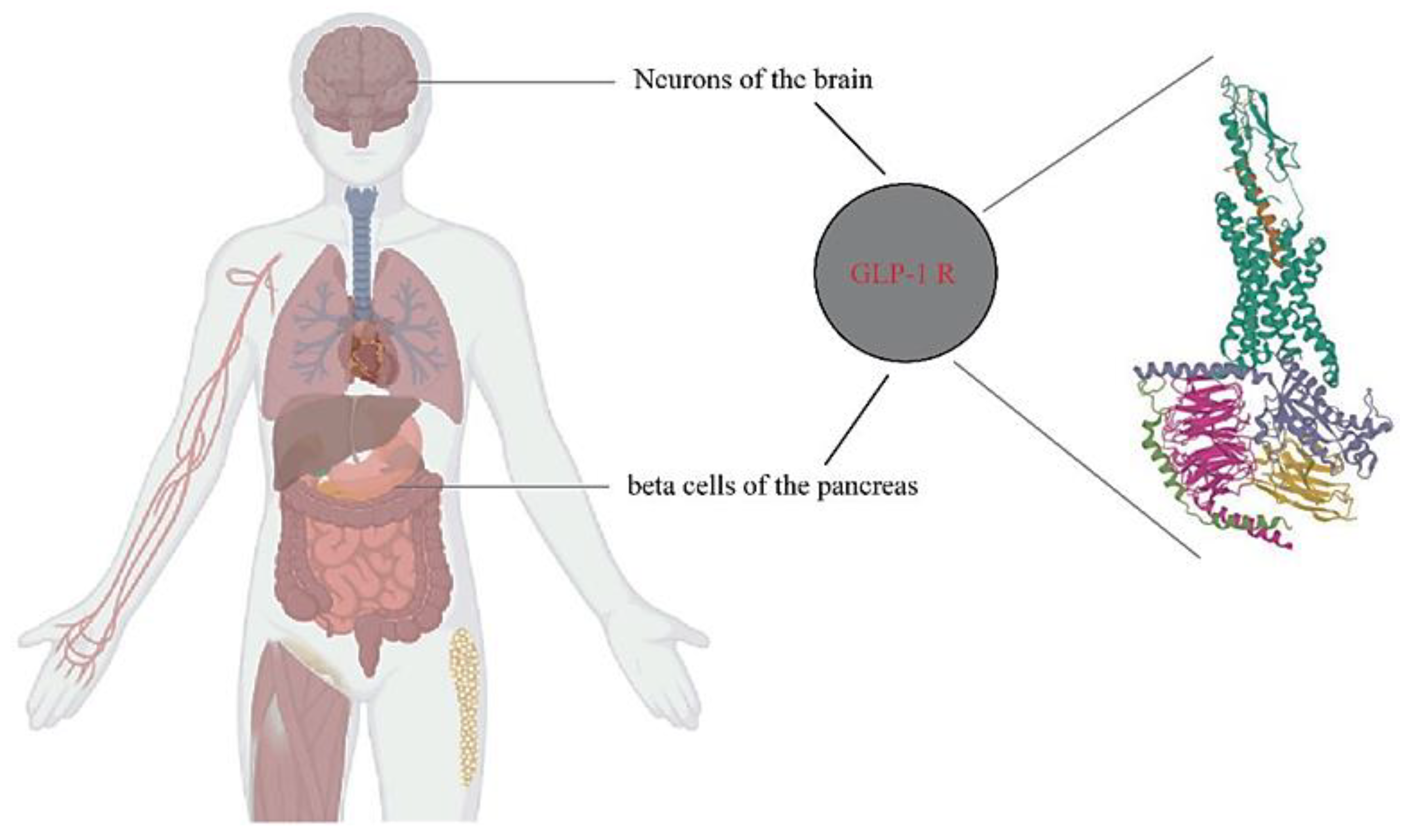
Table 5. Summary of clinical trials that investigated weight loss using Exenatide. Copyright: Simona Cavalu et al.
| Study | Dosage/Patients/Duration | Main Outcomes, Weight Reduction | Side Effects | Ref. |
|---|---|---|---|---|
| Duration-1 | Exenatide 2 mg/week, against the pre-existing 10 µg/twice per day version N = 295, 30 weeks | No increased risk of hypoglycaemia and similar reductions in body weight | Nausea reported in both treatments, but more often for 10 µg/twice per day formulation | [136] |
| Duration-2 | Exenatide (2 mg once/week) versus maximum approved doses sitagliptin, thiazolidinedione, or pioglitazone, in patients treated with metformin | Average 2.3 kg weight loss in exenatide group, 0.8 kg reduction in sitagliptin group, and 2.8 kg weight gain with pioglitazone | Nausea and diarrhea in exenatide and sitagliptin groups | [137] |
| Duration-3 | Exenatide (2 mg once/week) versus insulin glargine titrated to glucose targets N = 456, 84 weeks | Average 2.6 kg decrease in bodyweight with exenatide, compared with a 1.4 kg increase with glargine, accompanied by improved glycemic control | No evidence | [145] |
| Duration-4 | Exenatide once weekly (EQW) compared with metformin, pioglitazone, and sitagliptin (SITA) N = 820, 26 weeks | 2.0 kg decrease with exenatide versus 0.8 kg reduction with sitagliptin and 1.5 kg increase with pioglitazone | Exenatide once weekly induced nausea and diarrhea | [146] |
| Duration-5 | Exenatide (2 mg once/week)) versus exenatide twice daily (5 µg during 4 weeks followed by 10 µg during 20 weeks) in order to improve glycemic control, body weight, and safety. N = 252, 24 weeks | Similar reductions in mean body weight from baseline to wk 24 observed in both groups (−2.3 ± 0.4 kg and −1.4 ± 0.4 kg) | In both groups, the majority of nausea was transient and mild to moderate in intensity, while the incidence decreased over time | [138] |
| Duration-6 | Exenatide once weekly (2 mg) versus liraglutide (1.8 mg) once daily in patients with T2DM. N = 911, 26 weeks | Better body weight reductions in liraglutide group (average 2.68–3.57 kg) | Nausea predominantly in exenatide group; diarrhea and vomiting more frequently in the liraglutide group and with decreasing incidence over time in both groups | [144] |
| Duration-7 | Exenatide 2 mg once weekly or placebo in patients with T2DM inadequately controlled despite titrated insulin glargine ± metformin. N = 461, 28 weeks | Body weight reduction average of 1.5 kg with exenatide versus placebo. | Gastrointestinal and injection-site adverse events more frequent with exenatide + IG than with placebo + IG | [140] |
| Duration Neo-1 | Exenatide 2 mg once/week, self-injectable Miglyol suspension (QWS-AI) versus exenatide 10 µg twice daily (BID), N = 375, 28 weeks | Significant body weight was reduced in both groups | Gastrointestinal adverse events were reported in 22.7% of patients within exenatide QWS-AI group and 35.6% in exenatide BID group | [141,142] |
| Duration-Neo-2 | Exenatide 2 mg once-weekly Miglyol suspension for autoinjection (QWS-AI) versus sitagliptin (100 mg once/day oraly) or placebo. N = 364, 28 weeks | Average 1.12 kg and, respectively, 1.19 kg decrease of bodyweight in exenatide and sitagliptin groups versus 0.15 kg increase in the placebo | Gastrointestinal events and injection-site reactions | [142] |
In most studies, GLP-1 levels were not related to insulin concentration or measures of insulin resistance. In preclinical models, GLP-1 mainly demonstrates a stimulatory effect on the HPG axis. Therefore, pharmacological stimulation of the GLP-1R by GLP-1RA might be able to reverse gonadotropin suppression in various states of metabolic imbalance.Due to the complexity of biological systems, the final effect of GLP-1 on the HPG axis is multifactorial and appears to integrate other synergistic and counterbalancing metabolic and endocrine factors. In addition, GLP-1 appears to have a direct anti-fibrotic and anti-inflammatory effect on peripheral reproductive tissues.In conclusion, clinical studies and the anatomical distribution of GLP-1R suggest that GLP-1 might play a vital role as a modulatory signal between metabolic and reproductive systems. Management of comorbidities increasingly common in T2DM patients, such as obesity and liver disease, needs to be better addressed. In this regard, ongoing studies will provide further information on whether the benefits of GLP-1 extend to these indications. Copyright: Simona Cavalu et al.
The full text here: https://www.mdpi.com/1422-0067/24/13/10449
